
The ions are then separated in a mass analyzer according to their mass-to-charge ratio. It requires that all molecules and atoms to be measured be ionized. Mass spectrometry is a technique that measures the mass-to-charge ratio (m/q) of ions. Masses of atoms and molecules are measured by mass spectrometry. Neither u nor Da are SI units, but both are recognized by the SI. For this reason, the dalton (Da) is increasingly recommended as the accurate mass unit. Hence, the amu is no longer in use those who still use it do so with the definition of the u in mind. Therefore, both communities agreed to the compromise of using m( 12C)/12 as the new unit, naming it the "unified atomic mass unit" (u). Because the isotopic distribution in nature can change, this definition is a moving target. The amu was defined differently by physicists and by chemists:Ĭhemists used oxygen in the naturally occurring isotopic distribution as the reference. Some chemists use the atomic mass unit (amu). Berzelius demonstrated that this is not always the case by showing that chlorine (Cl) has a mass of 35.45, which is not a whole number multiple of hydrogen's mass. Known was Prout's Law, Prout suggested that the known elements had atomic weights that were whole number multiples of the atomic mass of hydrogen. Early atomic mass theory was proposed by the English chemist William Prout in a series of published papers in 18. The first scientists to measure atomic mass were John Dalton (between 18) and Jons Jacoband Berzelius (between 18). So 1 u is 1/12 of the mass of a carbon-12 isotope: Both units are derived from the carbon-12 isotope, as 12 u is the exact atomic mass of that isotope. The atomic mass is usually measured in the units unified atomic mass unit (u), or dalton (Da). These concepts are further explained below. Mass of molecule calculated from the mass of its isotopes (in contrast of measured ba a mass spectrometer) Integer mass of molecule consisting of most abundant isotopes Ratio of mass m of a molecule and and the atomic mass constant m u
Ratio of mass m and and the atomic mass constant m u Mass Concepts in Chemistry name in chemistry Note that the former is now often referred to as the "molecular weight" or "atomic weight". The former usually implies a certain isotopic distribution, whereas the latter usually refers to the most common isotope ( 16O 2). The molar mass of the N 2 molecule is therefore 28.02 g/mol. This is referred to as the molecular mass and the molecular mass of any molecule is simply the sum of the atomic masses of all of the elements in that molecule. For example, the macroscopic mass of oxygen (O 2) does not correspond to the microscopic mass of O 2. For nitrogen, the mass of the N 2 molecule is simply (14.01 + 14.01) 28.02 amu. This means that from a physical stand point, these mixtures are not pure. On the macroscopic level, most mass measurements of pure substances refer to a mixture of isotopes. In addition, the situation is rendered more complicated by the isotopic distribution. The name "atomic mass" is used for historical reasons, and originates from the fact that chemistry was the first science to investigate the same physical objects on macroscopic and microscopic levels.

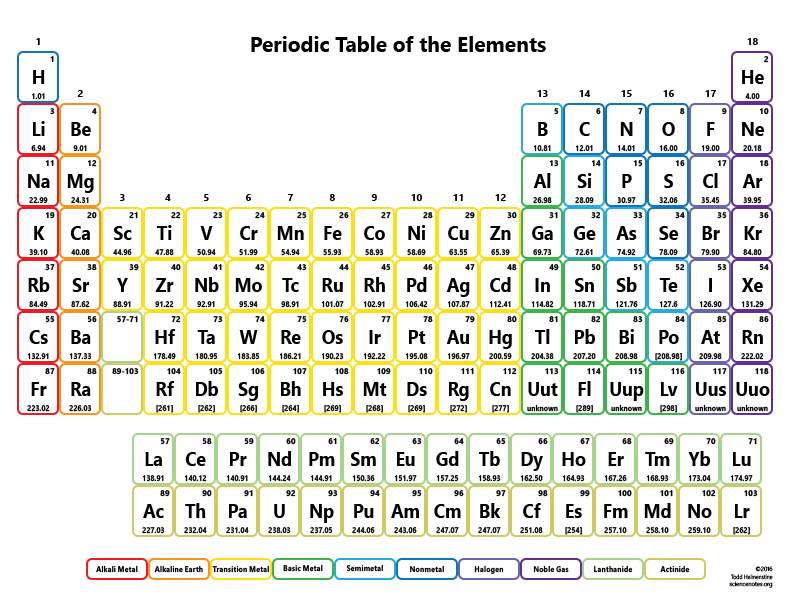
"Neither the name of the physical quantity, nor the symbol used to denote it, should imply a particular choice of unit." Hint: Count the number of atoms of each element, and then multiply that number by the element's atomic weight.\) Use the table below to find the atomic weight of each atom (element), or refer to a Periodic Table of the Elements. Let us calculate the molecular weight of some common compounds. For example, in one mole of a chemical compound there are 6.022 x 1023 molecules. One mole of 'something' contains 6.022 x 1023 entities. A mole is the unit that measures the amount of a substance. One thousand mers connected together would add up to a weight of 28,000 grams/mole and would have 6,000 atoms.Ī mole is the standard method in chemistry for communicating how much of a substance is present. We combine (react) many mers of ethylene together to form a polyethylene chain. The atomic weight of carbon is 12 and that of hydrogen is 1, so one mer of ethylene has a weight of 2(12) + 4(1) = 28. It has a total of 6 atoms: 2 carbon (C) atoms and 4 hydrogen (H) atoms. The chemical formula for an ethylene monomer is -(CH2-CH2).In order to calculate the molecular weight of one water molecule, we add the contributions from each atom that is, 2(1) + 1(16) = 18 grams/mole. Using the periodic table of the elements to find atomic weights, we find that hydrogen has an atomic weight of 1, and oxygen's is 16. The chemical formula for water is H2O, which means this molecule has 3 atoms: 2 of hydrogen (H) and 1 oxygen (O) atom.
